
healthy.io made two important announcements yesterday, first that they had received FDA approval for their smartphone based production to make take and ACR test to help diagnose chronic kidney disease which affects over 35 million Americans. T
Second the company announced that it had closed a $60 million Series C funding round led by Corner Ventures with participation by Joy Capital and all previous investors: Ansonia Holdings, Aleph, and Samsung NEXT.
The funding round will be used to accelerate Healthy.io’s global expansion and product development. Additionally, the company announced that longtime leader in health care innovation, former executive vice president of Aetna Gary Loveman, Ph.D. will join its Board of Directors.
“The smartphone has the potential to be the great equalizer of healthcare,” said Yonatan Adiri, founder and CEO of Healthy.io. “To unlock this potential, companies must adhere to the highest clinical standards. Our second FDA clearance and additional funding allows us to expand access to critical tests and care beyond our first 100,000 patients in Europe and Israel, making it a reality for millions of Americans suffering from diabetes and hypertension, who are at risk for chronic kidney disease.”
The FDA clearance designates Healthy.io’s smartphone-based ACR test as substantially equivalent to lab-based testing and authorizes the use of the test by healthcare professionals at any point of care. It makes it possible for any pharmacy, urgent care center, or health clinic to perform the test without investing in a tabletop lab device. In addition, Healthy.io’s solution allows immediate electronic medical record (EMR) connectivity through the automated smartphone scan.
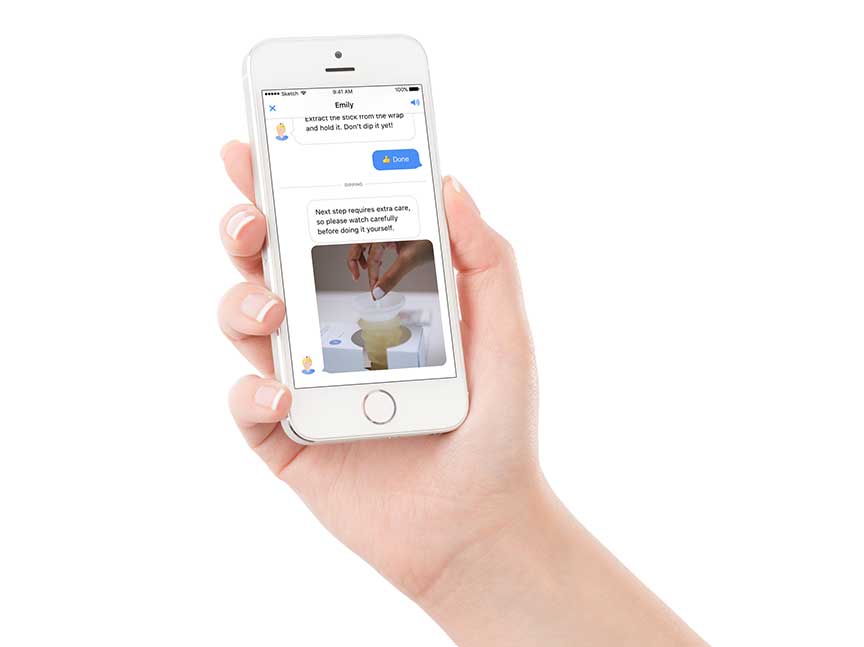
Last year, the company received clearance for its at-home, smartphone-based 10 parameter urinalysis test kit, called Dip.io, that can be used in testing for UTIs or in prenatal care. Next, the company plans to continue the approval process for its ACR test kit for at-home use.

